- Start by preparing individual single color controls for each marker in your panel. Ideally, the control should contain both a negative and positive population. If the marker is expressed on 100% of the cells, an unstained universal negative sample needs to be employed. If the marker is, for example, staining a cytokine or another marker not always expressed, the positive control should be done on cells stimulated to produce that expression.
- Ensure that the positive signal in your compensation control is at least as bright as your samples. If you are looking at a rare marker, you may want to consider using compensation beads instead of cells.
- Use the same fluorophore for compensation as you do to stain your samples. Using the same reagents for compensation is particularly important when staining with tandem dyes (such as PE/Cyanine7) due to potential lot-to-lot variation in the transfer of energy between the donor and acceptor molecules. Learn more about tandem dyes.
- Ensure that the autofluorescence of the negative and positive population are the same. If all of your compensation controls are stained using beads, the beads will all have the same degree of autofluorescence. You can use both beads and cells to compile your compensation profile; however, you can not choose the option to employ a universal negative control because the cells and beads will not have the same level of autofluorescence.
- Keep your PMT voltages as balanced as possible. The absolute compensation values are not only dependent on the spectral overlap between two fluorophores but also the PMT voltage. Remember the default CST settings may not be optimal for your sample, instead, use stained cells to adjust the PMT voltages for your experiment. To maintain balanced PMT voltages, it is important to build a balanced panel. For more information on building a balanced panel, check out our Multicolor Staining Guide.
- Treat your compensation controls and your samples the same way. For example, if you are fixing your samples, perform fixation on your compensation controls as well.
- Collect enough events to accurately calculate an MFI for the positive and negative population.
- Apply compensation to all parameters in your experiment. Remember some fluorophores can be minimally excited by multiple lasers which may cause spillover into channels that are not excited by the same laser.
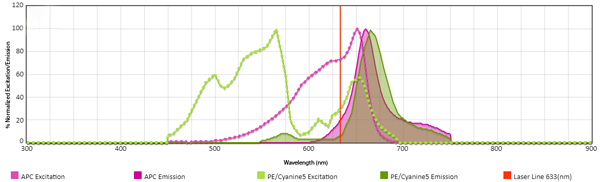
Using a Spectra Analyzer, you can see that the PE/Cyanine5 can be excited by the 633 nm laser, making it difficult to compensate the spillover into APC.
- Run freshly stained compensation controls for each experiment. Proper controls should be set up for each experiment, even if you've already generated a similar compensation matrix in the past.
- If you are using compensation beads, ensure that they will bind the isotype of your antibody of interest. Some compensation beads are specific to certain species or kappa light chains.
|

 Login/Register
Login/Register 







Follow Us